Catalysing the performance of Li–sulfur batteries with two-dimensional conductive metal organic frameworks†
Abstract
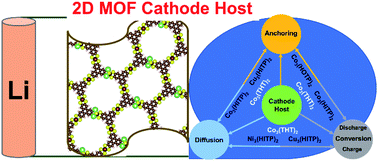
At present, the efficiency of high energy density Li–sulfur (Li–S) batteries is being narrowed by the limited strategies to deal with the shuttling effect, sluggish redox kinetics and poor ionic and electron conductivities of cathode hosts. Recently, synthesized triphenylene based two-dimensional conductive metal organic frameworks (2D MOFs) have been observed to hold promising characteristics to overcome the limitations with suitable pore and anchoring sites along with the desired electron conductivity. In this study, we use density functional theory calculations to investigate 2D honeycomb MOFs with hexa-substituted triphenylene linkers with pairs of varying metal ions (Cu, Ni, and Co) and functional groups (N, O, and S) (M3(2,3,6,7,10,11-hexaiminotriphenylene)2 (M3(HITP)2), M3(2,3,6,7,10,11-hexahydroxytriphenylene)2 (M3(HOTP)2) and M3(triphenylene-2,3,6,7,10,11-hexathiol)2 (M3(THT)2)) to be the potential cathode hosts in Li–S batteries and analyse their structure–activity relationship. The detailed study of structural-electronic characteristics followed by analysis of the triangle of catalytic activity concludes that M3(HOTP)2 can deliver higher anchoring efficiency, but it suffers from low redox reactivity with high Li diffusion and Li2S decomposition barriers, whereas M3(HITP)2 MOFs appear insubstantial with the possibility of Li polysulfide shuttling due to the lower anchoring. Co3(THT)2 not only has superior anchoring efficiency (−1.40 to −2.68 eV) but also shines in terms of facile Li+ diffusion (0.68 eV), a small Gibbs free energy requirement of 0.68 eV for Li polysulfide reduction and a moderately low Li2S decomposition barrier (1.06 eV) that simultaneously contribute to the performance of Li–S batteries. Our study concludes that the combination of Co and S in Co3(THT)2 makes the most suitable cathode host and provides a basis for the designing and screening of new materials having such active components to achieve high performance Li–S batteries.


 Please wait while we load your content...
Please wait while we load your content...