Quantifying the effects of cooperative hydrogen bonds between vicinal diols on polymer dynamics†
Abstract
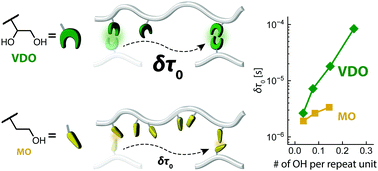
Transient cross-links such as hydrogen bonds (H-bonds) are a central concept for creating polymers with mechanical functionalities, including toughness and self-healing properties. While conventional strong H-bonding groups are based on rigid and planar molecular motifs with multidentate intermolecular interactions, we recently discovered that a structurally simple and flexible vicinal diol (VDO) could serve as a robust yet dynamic cross-link with multiple intermolecular H-bonds between hydroxy groups. In this work, we investigated the effects of cooperativity of H-bonds in VDOs on polymer dynamics. We synthesized model polybutadienes with either VDO or monool (MO) side groups by a radical-mediated thiol–ene click reaction. The oscillatory shear rheology data were analyzed by using the sticky Rouse model. The characteristic time of a single modified segment (δτ0) was significantly longer for the VDO-modified polymers than for the MO-modified polymers, even when they had the same number density of hydroxy groups. The increase in δτ0 with increasing degree of modification was much more drastic for the VDO-modified polymers than for the MO-modified polymers. Moreover, the characteristic time of an unmodified Rouse segment (τ0) was found to increase upon increasing the number of VDOs in the chain, while it was unchanged against the number of MOs. These observations highlight the cooperative effects of placing two hydroxy groups in a close vicinal arrangement. The multiplicity of H-bonds and the structural flexibility of VDOs led to efficient retardation of the chain dynamics.


 Please wait while we load your content...
Please wait while we load your content...