Complementary metal–semiconductor interactions on Ag-TiO2 NT heterojunction for the efficient electrochemical reduction of 5-hydroxymethylfurfural†
Abstract
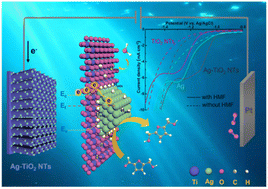
Electrocatalytic conversion of biomass-derived platform molecules is regarded as an emerging approach to obtain high-value fuels and chemicals. Herein, a heterostructured design strategy involving dispersed Ag nanoparticles immobilized on one-dimensional TiO2 nanotubes (Ag-TiO2 NTs) for the electroreduction of 5-hydroxymethylfurfural (HMF) into 2,5-dihydroxymethylfuran (DHMF) was reported. The optimal electrode exhibited an 82.2% FE, 89.8% selectivity, and DHMF productivity value of 0.1 mmol cm−2 h−1. Electrochemical measurements combined with density functional theory (DFT) calculations manifested that the superior performance was derived from the heterojunction-induced metal–semiconductor interactions at the interface, by which the TiO2 NTs and Ag bring complementary advantages inhibiting the hydrogen evolution reaction (HER) and facilitating charge transfer. Moreover, the catalytic activity of the Ag sites on Ag-TiO2 NTs was considered separately. The mass activity of Ag on Ag-TiO2 NTs reached 5.8 mol L−1 g−1, which was far more than that of Ag mesh (0.038 mol L−1 g−1), indicating the significantly enhanced activity of Ag with high utilization. This could be attributed to the controllable nucleation of Ag through the confinement effect of TiO2 NTs, acquiring dispersed Ag nanoparticles. The design scheme for the heterostructured Ag-TiO2 NTs electrocatalyst displays prospective applications for developing highly efficient electrocatalysts for the electrochemical reduction of HMF.


 Please wait while we load your content...
Please wait while we load your content...