Exploring the air stability of all-inorganic halide perovskites in the presence of photogenerated electrons by DFT and AIMD studies†
Abstract
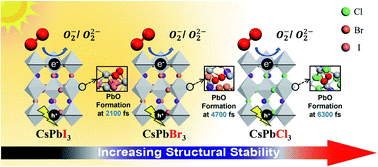
Developing ambient air and light stable metal halide perovskites could lead to tremendous progress in reducing energy waste and environmental damage through photocatalysis. In this work, we systematically investigated the structural stability of three all-inorganic lead halide perovskites (CsPbCl3, CsPbBr3, and CsPbI3) towards ambient air (O2); in particular, the role of light (photo-generated electrons and holes) in the perovskites' structural degradation was investigated using density functional theory (DFT) and ab initio molecular dynamics (AIMD) simulation methods. We find that the adsorbed O2 molecule is strongly activated on these perovskite surfaces even in the dark. Our results show that the presence of photo-generated electrons and holes increases the strength of O2 adsorption; specifically, photo-generated electrons enhance the electrostatic interactions of these perovskite surfaces with O2, resulting in a larger activation effect. AIMD simulations clearly show that adsorbed O2 is readily activated on the surfaces of CsPbBr3 and CsPbI3, and Pb–O bonds form spontaneously in the presence of photo-generated electrons. Although the CsPbCl3 perovskite surface has greater adsorption strength and activation effect towards O2 in the presence of photo-generated electrons, it does not undergo light-induced structural degradation. Based on these results, we suggest that CsPbCl3 perovskite can be a promising photocatalyst material.


 Please wait while we load your content...
Please wait while we load your content...