CoxNi1−xO-NiCo2O4/rGO synergistic bifunctional electrocatalysts for high-rate rechargeable zinc–air batteries†
Abstract
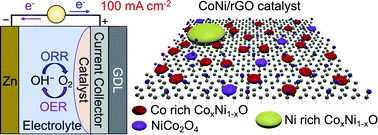
Bifunctional catalysts containing spinel NiCo2O4 and CoxNi1−xO bimetallic oxide nanoparticles embedded on reduced graphene oxide (rGO), CoNi/rGO, were obtained by the thermal decomposition of metal nitrates and graphene oxide (GO). For the first time, we report the correlation of the catalytic activities in the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) at low current density (<30 mA cm−2) and the zinc–air battery (ZAB) performance at a high current density of 100 mA cm−2 with the structures of CoNi/rGO catalysts with different Co : Ni molar ratios and metal loading. A layer of 2–3 nm NiCo2O4 and Co-rich CoxNi1−xO nanoparticles on rGO exhibited a synergistic ORR catalytic effect. In contrast, NiO and big-sized Ni-rich CoxNi1−xO nanoparticles (10–20 nm) exhibited poor ORR and OER performance at low current density. No synergetic effect of Co and Ni oxides were found in the OER at low current density in the electrochemical test. The battery test at high current density revealed the best cycling stability of CoNi/rGO-5/5 with equimolar Co and Ni and medium metal loading. This is due to the synergetic OER performance with the lowest charge voltage at high current density and the best ORR activity with the highest electron transfer numbers of 3.63. This is related to the highest Co : Ni ratio of the small-sized CoxNi1−xO bimetallic oxide and the high amount of spinel NiCo2O4 nanoparticles. Corrosion during charging was found to be the limiting factor of the long-term cycling stability at high current density.


 Please wait while we load your content...
Please wait while we load your content...