Improving the electron transfer in the oxygen reduction reaction by N/S co-doping for high-performance of Zn–air batteries†
Abstract
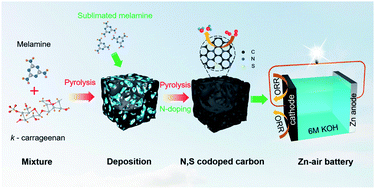
Designing high-performance carbon-based oxygen reduction catalysts using a facile strategy is an attractive research direction in the field of energy conversion and storage. Herein, N/S co-doped carbon was prepared using κ-carrageenan and melamine as precursors, in which melamine can be decomposed into nitrogen-containing gas for realizing nitrogen doping, and the sulfate groups in κ-carrageenan can be converted for sulfur doping. Such an N/S doping strategy enables a carbon catalyst with a large specific surface area, hierarchical porous structure, and rich defective sites to be achieved, displaying a high positive onset potential (Eonset = 0.98 V) and half wave potential (E1/2 = 0.89 V), excellent stability, and methanol tolerance. More importantly, S doping could increase the positive charge density of the carbon adjacent to the nitrogen, which is favorable for enhancing the chemisorption of O2 and weakening the O–O bond, thereby promoting the reaction from O2 to OH− and improving electron transfer. Furthermore, with this catalyst as the air cathode, the assembled aqueous Zn–air battery exhibits a high open-circuit potential of 1.53 V and a remarkable peak power density of 166.05 mW cm−2, outperforming the Pt/C catalyst. This work gives a facile and effective strategy to construct high-performance ORR catalysts with four electron transfer for Zn–air batteries.


 Please wait while we load your content...
Please wait while we load your content...