One-step construction of Y, C, and O tridoped g-C3N4 as a bifunctional photocatalyst for H2 evolution and organic pollutant degradation under visible light irradiation†
Abstract
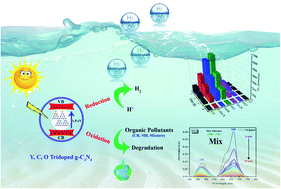
Photocatalytic H2 production and degradation of pollutants are promising strategies for energy conversion and environmental protection, where highly efficient photocatalysts are required. Herein, a novel type of Y, C, and O tridoped g-C3N4 bifunctional photocatalyst was prepared via a simple one-step low-cost thermal polymerization technique, which exhibits a significant enhancement in photocatalytic redox efficiency under visible light for H2 production and degradation of pollutants compared to bulk g-C3N4. The optimized Y0.1/C/O tridoped g-C3N4 photocatalyst exhibits a remarkable H2 evolution rate (HER = 2542.4 μmol g−1 h−1) which is 5 times higher than that of pristine g-C3N4 with a high apparent quantum yield of 6% at 420 nm, besides excellent degradation efficiencies for organic pollutants (∼100, 98.5 and 82.4% for mixed dyes, congo red, and methylene blue, respectively). The influence of Y3+ concentration on the photocatalytic performance and electronic structure of Y/C/O–CN was also investigated. The major reactive species involved in the photodegradation process were found to be superoxide (O2˙−) radicals. Moreover, the experimental and computational results suggest that the enhanced photocatalytic performance is due to the synergistic effect of Y, C, and O tridoping, which can adjust the band structure of g-C3N4, reduce the bandgap, improve visible-light absorption, and accelerate charge separation. This study paves the way to fabricate extremely effective tridoped photocatalysts for efficiently evolving hydrogen under visible light and resolving future environmental pollution.


 Please wait while we load your content...
Please wait while we load your content...