Lattice-disorder layer generation from liquid processing at room temperature with boosted nanointerface exposure toward water splitting†
Abstract
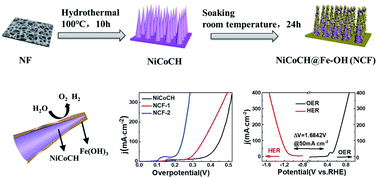
The structural symmetry breaking of a highly crystalline substance towards a chaotic non-crystal phase construction, particularly involving a self-healing process after crystalline grain destruction, is beset by difficulties and is critical for high-performance water splitting. Taking CO32−-intercalated carbonate hydroxides as initial high-crystallinity crystals, controllable interfacial structure destruction was designed on the basis of surface-wetting invasion from Fe relevant Lewis-acidic metal salts. A spontaneous self-treatment course occurs via the in situ formation of an ultrathin lattice-disordered layer (∼7.5 nm in thickness) due to the interfacial hydrolysis of the escaped CO32− encountering positive-valence iron ions. As-obtained NiCoCH@Fe–OH, with Co(CO3)0.5(OH)·0.11H2O@Ni2(OH)2CO3 (NiCoCH) as the core and amorphous Fe(OH)3 as the shell, shows excellent catalytic performance toward water splitting, with a current density of 50 mA cm−2 at overpotentials of 221 mV for the oxygen evolution reaction and 233.8 mV for the hydrogen evolution reaction in 1.0 M KOH. Assembling a two-electrode alkaline electrolytic cell, only 1.69 V is required to drive a current density of 50 mA cm−2. This work merges structural destruction and in situ regeneration into a one-material system through a single-step surface-wetting strategy for precise surficial nanostructure design, and it displays a mild/scalable approach for high-activity catalyst development.


 Please wait while we load your content...
Please wait while we load your content...