Leveraging coordination chemistry in the design of bipolar energy storage materials for redox flow batteries†
Abstract
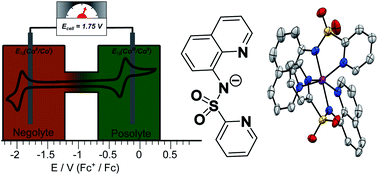
Tuned by the ligand field, some single metal atoms in coordination complexes can offer two redox processes with a large potential difference, a key characteristic of symmetrical redox flow batteries (RFBs). We have designed a new anionic tridentate ligand N-(quinolin-8-yl)pyridine-2-sulfonamide (psq−) with the aim of tuning the CoIII/CoII and CoII/CoI couples compared to those for analogous bis-homoleptic cobalt complexes of 2,2′:6′,2′′-terpyridine (terpy) and derivatives which have been tested as bipolar energy storage molecules. With similar structural parameters to these bis-terpy analogues, the single crystal X-ray structures of [M(psq)2] (M = CoII and ZnII) and [CoIII(psq)2](PF6) show a planar ligand geometry. However the cyclic voltammogram of CoII(psq)2 reveals negative shifts of 0.840 V and 0.175 V for the reversible CoII/CoI and CoIII/CoII redox processes compared to the bis-terpy analogue. Therefore, with a peak to peak separation between the CoII/CoI and CoIII/CoII couples that is 0.68 V larger, bipolar RFBs employing CoII(psq)2 as the energy storage material will operate at higher cell potentials than those offered by [CoII(terpy)2]2+. In an H-cell set-up, CoII(psq)2 delivers an initial effective charge to 83%. A charge capacity fade of 3% per cycle observed over 30 cycles is partly ascribed to ligand reduction. This work shows that psq− is a promising scaffold for bipolar energy storage molecules and illustrates the opportunities for innovation in RFB design through the development of fit-for-purpose redox-active coordination complexes.


 Please wait while we load your content...
Please wait while we load your content...