Buffering the local pH via single-atomic Mn–N auxiliary sites to boost CO2 electroreduction†
Abstract
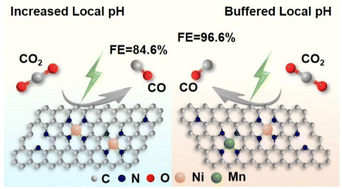
Electrocatalytic CO2 reduction driven by renewable energy has become a promising approach to rebalance the carbon cycle. Atomically dispersed transition metals anchored on N-doped carbon supports (M-N-C) have been considered as the most attractive catalysts to catalyze CO2 to CO. However, the sluggish kinetics of M-N-C limits the large-scale application of this type of catalyst. Here, it is found that the introduction of single atomic Mn–N auxiliary sites could effectively buffer the locally generated OH− on the catalytic interface of the single-atomic Ni–N–C sites, thus accelerating proton-coupled electron transfer (PCET) steps to enhance the CO2 electroreduction to CO. The constructed diatomic Ni/Mn–N–C catalysts show a CO faradaic efficiency of 96.6% and partial CO current density of 13.3 mA cm−2 at −0.76 V vs. RHE, outperforming that of monometallic single-atomic Ni–N–C or Mn–N–C counterparts. The results suggest that constructing synergistic catalytic sites to regulate the surface local microenvironment might be an attractive strategy for boosting CO2 electroreduction to value-added products.


 Please wait while we load your content...
Please wait while we load your content...