Thiazole fused S,N-heteroacene step-ladder polymeric semiconductors for organic transistors†
Abstract
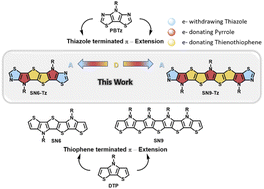
Ladder-type thiazole-fused S,N-heteroacenes with an extended π-conjugation consisting of six (SN6-Tz) and nine (SN9-Tz) fused aromatic rings have been synthesized and fully characterized. To date, the synthesis of well-defined fused building blocks and polymers of π-conjugated organic compounds based on the thiazole moiety is a considerable synthetic challenge, due to the difficulty in their synthesis. Acceptor–donor building blocks M1 and M2 were successfully polymerized into ladder homopolymers P1–P2 and further copolymerized with a diketopyrrolopyrrole unit to afford step-ladder copolymer P3. The optical, electronic, and thermal properties, in addition to their charge transport behavior in organic thin-film transistors (OTFTs), were investigated. The results showed an interesting effect on the molecular arrangement of the thiazole-based ladder-type heteroacene in the crystal structure revealing skewed π–π-stacking, and expected to possess better p-type semiconducting performance. The polymers all possess good molecular weights and excellent thermal properties. All the polymer-based OTFT devices exhibit annealing temperature dependent performance, and among the polymers P3 exhibits the highest mobility of 0.05 cm2 V−1 s−1.
- This article is part of the themed collection: #RSCPoster Conference


 Please wait while we load your content...
Please wait while we load your content...