Electrochemical oxidative rearrangement of tetrahydro-β-carbolines in a zero-gap flow cell†
Abstract
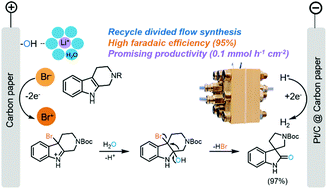
Oxidative rearrangement of tetrahydro-β-carbolines (THβCs) is one of the most efficient methods for the synthesis of biologically active spirooxindoles, including natural products and drug molecules. Here, we report the first electrochemical approach to achieve this important organic transformation in a flow cell. The key to the high efficiency was the use of a multifunctional LiBr electrolyte, where the bromide (Br−) ion acts as a mediator and catalyst and lithium ion (Li+) acts as a likely hydrophilic spectator, which might considerably reduce diffusion of THβCs into the double layer and thus prevent possible nonselective electrode oxidation of indoles. Additionally, we build a zero-gap flow cell to speed up mass transport and minimize concentration polarization, simultaneously achieving a high faradaic efficiency (FE) of 96% and an outstanding productivity of 0.144 mmol (h−1 cm−2). This electrochemical method is demonstrated with twenty substrates, offering a general, green path towards bioactive spirooxindoles without using hazardous oxidants.


 Please wait while we load your content...
Please wait while we load your content...