Activation of MoS2 monolayer electrocatalysts via reduction and phase control in molten sodium for selective hydrogenation of nitrogen to ammonia†
Abstract
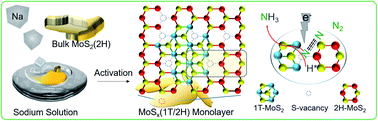
Electrochemical nitrogen fixation under ambient conditions is promising for sustainable ammonia production but is hampered by high reaction barrier and strong competition from hydrogen evolution, leading to low specificity and faradaic efficiency with existing catalysts. Here we describe the activation of MoS2 in molten sodium that leads to simultaneous formation of a sulfur vacancy-rich heterostructured 1T/2H-MoSx monolayer via reduction and phase transformation. The resultant catalyst exhibits intrinsic activities for electrocatalytic N2-to-NH3 conversion, delivering a faradaic efficiency of 20.5% and an average NH3 rate of 93.2 μg h−1 mgcat−1. The interfacial heterojunctions with sulfur vacancies function synergistically to increase electron localization for locking up nitrogen and suppressing proton recombination. The 1T phase facilitates H–OH dissociation, with S serving as H-shuttling sites and to stabilize 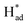 . The
. The 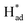 subsequently couple with nearby N2 and NHx intermediates bound at Mo sites, thus greatly promoting the activity of the catalyst. First-principles calculations revealed that the heterojunction with sulfur vacancies effectively lowered the energy barrier in the potential-determining step for nitrogen reduction, and, in combination with operando spectroscopic analysis, validated the associative electrochemical nitrogen reduction pathway. This work provides new insights on manipulating chalcogenide vacancies and phase junctions for preparing monolayered MoS2 with unique catalytic properties.
subsequently couple with nearby N2 and NHx intermediates bound at Mo sites, thus greatly promoting the activity of the catalyst. First-principles calculations revealed that the heterojunction with sulfur vacancies effectively lowered the energy barrier in the potential-determining step for nitrogen reduction, and, in combination with operando spectroscopic analysis, validated the associative electrochemical nitrogen reduction pathway. This work provides new insights on manipulating chalcogenide vacancies and phase junctions for preparing monolayered MoS2 with unique catalytic properties.


 Please wait while we load your content...
Please wait while we load your content...