A switchable redox annulation of 2-nitroarylethanols affording N-heterocycles: photoexcited nitro as a multifunctional handle†
Abstract
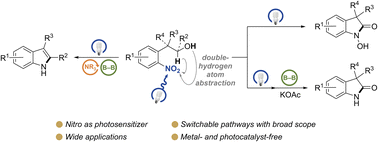
The efficient transformation of nitroaromatics to functional molecules such as N-heterocycles has been an attractive and significant topic in synthesis chemistry. Herein, a photoexcited nitro-induced strategy for switchable annulations of 2-nitroarylethanols was developed to construct N-heterocycles including indoles, N-hydroxyl oxindoles and N–H oxindoles. The metal- and photocatalyst-free reaction proceeds through intramolecular redox C–N coupling of branched hydroxyalkyl and nitro units, which is initiated by a double hydrogen atom abstraction (d-HAA) process. The key to the switchable reaction outcomes is the mediation of a diboron reagent by its favorable oxy-transfer reactivity to in situ generated nitroso species. The utility of this protocol was well demonstrated by broad substrate scope, excellent yields, functional group tolerance and wide applications. Finally, detailed mechanistic studies were performed, and kinetic isotope effect (KIE) experiments indicate that the homolysis of the C–H bond is involved in the rate-determining step.
- This article is part of the themed collection: 2022 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...