The effect of specific adsorption of halide ions on electrochemical CO2 reduction†
Abstract
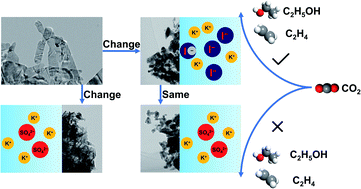
In the electrochemical CO2 reduction reaction (CO2RR), halide ions could impose a significant effect on multi-carbon (C2+) product production for Cu-based catalysts by a combined contribution from various mechanisms. However, the nature of specific adsorption of halide ions remains elusive due to the difficulty in decoupling different effects. This paper describes a facile method to actively immobilize the morphology of Cu-based catalysts during the CO2RR, which makes it possible to reveal the fundamental mechanism of specific adsorption of halide ions. A stable morphology is obtained by pre-reduction in aqueous KX (X = Cl, Br, I) electrolytes followed by conducting the CO2RR using non-buffered and non-specifically adsorbed K2SO4 as the supporting electrolyte, by which the change of local pH and cation concentration is also maintained during the CO2RR. In situ spectroscopy revealed that the specific adsorption of halide ions enhances the adsorption of *CO intermediates, which enables a high selectivity of 84.5% for C2+ products in 1.0 M KI.


 Please wait while we load your content...
Please wait while we load your content...