Total synthesis of Lentinus giganteus glycans with antitumor activities via stereoselective α-glycosylation and orthogonal one-pot glycosylation strategies†
Abstract
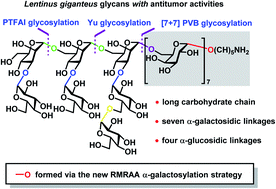
The accessibility to long, branched and complex glycans containing many 1,2-cis glycosidic linkages with precise structures remains a challenging task in chemical synthesis. Reported here is an efficient, stereoselective and orthogonal one-pot synthesis of a tetradecasaccharide and shorter sequences from Lentinus giganteus polysaccharides with antitumor activities. The synthetic strategy consists of: (1) newly developed merging reagent modulation and remote anchimeric assistance (RMRAA) α-(1→6)-galactosylation in a highly stereoselective manner, (2) DMF-modulated stereoselective α-(1→3)-glucosylation, (3) RMRAA stereoselective α-(1→6)-glucosylation, (4) several orthogonal one-pot glycosylations on the basis of N-phenyltrifluoroacetimidate (PTFAI) glycosylation, Yu glycosylation and ortho-(1-phenylvinyl)benzoate (PVB) glycosylation to streamline oligosaccharide synthesis, and (5) convergent [7 + 7] glycosylation for the final assembly of the target tetradecasaccharide. In particular, this new RMRAA α-galactosylation method has mild reaction conditions, broad substrate scopes and significantly shortened step counts for the heptasaccharide synthesis in comparison with 4,6-di-tert-butylsilyene (DTBS) directed α-galactosylation. Furthermore, DFT calculations shed light on the origins of remote anchimeric assistance effects (3,4-OBz > 3,4-OAc > 4-OBz > 3-OBz) of acyl groups.
- This article is part of the themed collection: 2022 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...