Photoredox catalysis via consecutive 2LMCT- and 3MLCT-excitation of an Fe(iii/ii)–N-heterocyclic carbene complex†
Abstract
Fe–N-heterocyclic carbene (NHC) complexes attract increasing attention as photosensitisers and photoredox catalysts. Such applications generally rely on sufficiently long excited state lifetimes and efficient bimolecular quenching, which leads to there being few examples of successful usage of Fe–NHC complexes to date. Here, we have employed [Fe(III)(btz)3]3+ (btz = (3,3′-dimethyl-1,1′-bis(p-tolyl)-4,4′-bis(1,2,3-triazol-5-ylidene))) in the addition of alkyl halides to alkenes and alkynes via visible light-mediated atom transfer radical addition (ATRA). Unlike other Fe–NHC complexes, [Fe(III/II)(btz)3]3+/2+ benefits from sizable charge transfer excited state lifetimes ≥0.1 ns in both oxidation states, and the Fe(III) 2LMCT and Fe(II) 3MLCT states are strong oxidants and reductants, respectively. The combined reactivity of both excited states enables efficient one-electron reduction of the alkyl halide substrate under green light irradiation. The two-photon mechanism proceeds via reductive quenching of the Fe(III) 2LMCT state by a sacrificial electron donor and subsequent excitation of the Fe(II) product to its highly reducing 3MLCT state. This route is shown to be more efficient than the alternative, where oxidative quenching of the less reducing Fe(III) 2LMCT state by the alkyl halide drives the reaction, in the absence of a sacrificial electron donor.
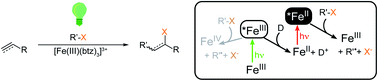
- This article is part of the themed collection: Most popular 2022 catalysis articles


 Please wait while we load your content...
Please wait while we load your content...