Iridium-catalyzed α-selective deuteration of alcohols†
Abstract
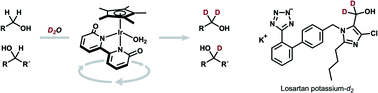
The development of chemoselective C(sp3)-H deuteration is of particular interest in synthetic chemistry. We herein report the α-selective, iridium(III)-bipyridonate-catalyzed hydrogen(H)/deuterium(D) isotope exchange of alcohols using deuterium oxide (D2O) as the primary deuterium source. This method enables the direct, chemoselective deuteration of primary and secondary alcohols under basic or neutral conditions without being affected by coordinative functional groups such as imidazole and tetrazole. Successful substrates for deuterium labelling include the pharmaceuticals losartan potassium, rapidosept, guaifenesin, and diprophylline. The deuterated losartan potassium shows higher stability towards the metabolism by CYP2C9 than the protiated analogue. Kinetic and DFT studies indicate that the direct deuteration proceeds through dehydrogenation of alcohol to the carbonyl intermediate, conversion of [IrIII–H] to [IrIII−D] with D2O, and deuteration of the carbonyl intermediate to give the α-deuterated product.
- This article is part of the themed collection: Most popular 2022 organic chemistry articles


 Please wait while we load your content...
Please wait while we load your content...