In situ Raman spectroscopy reveals the structure evolution and lattice oxygen reaction pathway induced by the crystalline–amorphous heterojunction for water oxidation†
Abstract
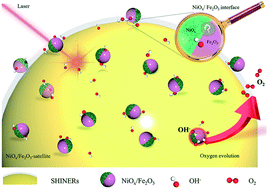
One of the most successful approaches for balancing the high stability and activity of water oxidation in alkaline solutions is to use amorphous and crystalline heterostructures. However, due to the lack of direct evidence at the molecular level, the nano/micro processes of amorphous and crystalline heterostructure electrocatalysts, including self-reconstruction and reaction pathways, remain unknown. Herein, the Leidenfrost effect assisted electrospray approach combined with phase separation was used for the first time to create amorphous NiOx/crystalline α-Fe2O3 (a-NiOx/α-Fe2O3) nanowire arrays. The results of in situ Raman spectroscopy demonstrate that with the increase of the potential at the a-NiOx/α-Fe2O3 interface, a significant accumulation of OH can be observed. Combining with XAS spectra and DFT calculations, we believe that more OH adsorption on the Ni centers can facilitate Ni2+ deprotonation to achieve the high-valence oxidation of Ni4+ according to HSAB theory (Fe3+ serves as a strong Lewis acid). This result promotes the electrocatalysts to follow the lattice oxygen activation mechanism. This work, for the first time, offers direct spectroscopic evidence for deepening the fundamental understanding of the Lewis acid effect of Fe3+, and reveals the synergistic effect on water oxidation via the unique amorphous and crystalline heterostructures.


 Please wait while we load your content...
Please wait while we load your content...