Radical boron migration of allylboronic esters†
Abstract
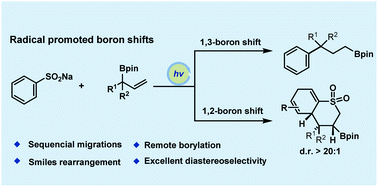
A photocatalyzed 1,3-boron shift of allylboronic esters is reported. The boron atom migration through the allylic carbon skeleton proceeds via consecutive 1,2-boron migrations and Smiles-type rearrangement to furnish a variety of terminally functionalized alkyl boronates. Several types of migrating variations of heteronuclei radicals and dearomatization processes are also tolerated, allowing for further elaboration of highly functionalized boron-containing frameworks.


 Please wait while we load your content...
Please wait while we load your content...