Ring-opening fluorination of bicyclic azaarenes†
Abstract
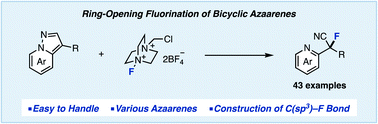
We have discovered a ring-opening fluorination of bicyclic azaarenes. Upon treatment of bicyclic azaarenes such as pyrazolo[1,5-a]pyridines with electrophilic fluorinating agents, fluorination of the aromatic ring is followed by a ring-opening reaction. Although this overall transformation can be classified as an electrophilic fluorination of an aromatic ring, it is a novel type of fluorination that results in construction of tertiary carbon–fluorine bonds. The present protocol can be applied to a range of bicyclic azaarenes, tolerating azines and a variety of functional groups. Additionally, mechanistic studies and enantioselective fluorination have been examined.


 Please wait while we load your content...
Please wait while we load your content...