Phenylene segments of zigzag carbon nanotubes synthesized by metal-mediated dimerization†
Abstract
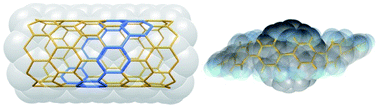
Well-studied cycloparaphenylenes (CPPs) correspond to the simplest segments of armchair CNTs, whereas the corresponding macrocyclic oligophenylene strip of zigzag CNTs is still missing. Herein, we present two series of conjugated macrocycles (CM2PP and CN2PP) containing two meta-phenylene or 2,7-naphthylene units facing each other in the strip. CM2PP and CN2PP can be regarded as the shortest cyclic primitive segments of zigzag CNTs. They were synthesized by gold-mediated dimerization and unambiguously characterized. They adopted the tubular structures and can further pack into one-dimensional supramolecular nanotubes. In particular, the supramolecular nanotube of CM2P4P mimics the CNT(9, 0) structure. Structural analysis and theoretical calculation accounted for the reduced ring strain in CM2PPs and CN2PPs. CM2PPs and CN2PPs exhibited a large optical extinction coefficient and high photoluminescence quantum yield. CN2P8P can accommodate fullerene C60, forming a Saturn-like C60@CN2P8P complex, a mimic structure of zigzag CNT peapods.


 Please wait while we load your content...
Please wait while we load your content...