Tunable heteroaromatic azoline thioethers (HATs) for cysteine profiling†
Abstract
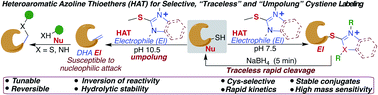
Here we report a new series of hydrolytically stable chemotype heteroaromatic azoline thioethers (HATs) to achieve highly selective, rapid, and efficient covalent labeling of cysteine under physiological conditions. Although the resulting cysteine–azoline conjugate is stable, we highlight traceless decoupling of the conjugate to afford unmodified starting components in response to reducing conditions. We demonstrated that HAT probes reverse the reactivity of nucleophilic cysteine to electrophilic dehydroalanine (Dha) under mild basic conditions. We demonstrated the umpolung capability of HAT probes for the modification of cysteine on peptides and proteins with various nucleophiles. We demonstrated that HAT probes increase the mass sensitivity of the modified peptides and proteins by 100 fold as compared to the classical methods. Finally, we extended the application of HAT probes for specific modification of cysteines in a complex cell lysate mixture.
- This article is part of the themed collection: Most popular 2021 chemical biology articles


 Please wait while we load your content...
Please wait while we load your content...