Pyrolytic activation of cellulose: energetics and condensed phase effects†
Abstract
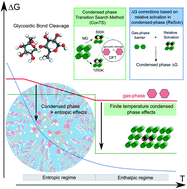
Bottom-up design of lignocellulose pyrolysis to optimize the quality and yield of bio-oil is hindered by the limited knowledge of the underlying condensed phase biomass chemistry. The influence of condensed phase and finite temperature effects on the energetics of glycosidic bond cleavage (a key step in cellulose breakdown) is investigated using two novel computational strategies and cellobiose as a model compound. Two temperature dependent reaction regimes were identified. Disruption of the hydrogen bonds at high temperatures provides additional conformational freedom making the glycosidic bond more accessible for protonation and in turn cleaves with a lower barrier. This is due to the relative destabilization of the reacting cellobiose molecule rather than the stabilization of the transition state. Such reactant destabilization and further enhancement of glycosidic bond activation by entropic contributions provide a novel outlook on cellulose chemistry in the condensed phase.


 Please wait while we load your content...
Please wait while we load your content...