Structure and dissolution of silicophosphate glass
Abstract
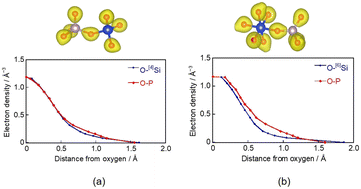
P2O5–SiO2–Na2O–CaO glasses are promising therapeutic ion-releasing materials. Herein, we investigated the state of silicon (Si) in P2O5–SiO2–Na2O–CaO glass using a model with a composition of 55.0P2O5–21.3SiO2–23.7Na2O (mol%), incorporating a six-fold-coordinated silicon structure ([6]Si). The model was constructed using a classical molecular dynamics method and relaxed using the first-principles method. Further, we experimentally prepared glasses, substituting Na2O for CaO, to investigate the dissolution of glass with varying [6]Si and PO4 tetrahedra (QPn) distributions (n = number of bridging oxygens (BOs) to neighboring tetrahedra). [6]Si in the glass model preferentially coordinated with QP3. When Si was surrounded by phosphate groups, phosphorus (P) induced the formation of [6]Si by elongating the Si–O distance, and [6]Si acted like a glass network former (NWF). Na+ coordinated with [6]Si–O–P bonds via electrostatic interactions with BO. 31P and 29Si magic-angle-spinning-nuclear-magnetic-resonance spectra of three experimental glass samples with the compositions of 55.0P2O5–21.3SiO2–xCaO–(23.7 − x)Na2O (mol%, x = 0, 12.4, and 23.7) showed that QP3 and [6]Si increased with increasing Na2O. When each glass powder was immersed in a tris-HCl buffer solution at 37 °C, the dissolution of NWF ions and network modifier (NWM) ions increased almost monotonically with time for all samples, indicating that the solubility of the samples was suppressed by the coexistence of CaO and Na2O, attributed to the delocalization of the electron distribution of P in the [6]Si-coordinated QP3 units compared to that in the P- or [4]Si-coordinated QP3 units, which reduces hydrolysis.


 Please wait while we load your content...
Please wait while we load your content...