Retracted Article: Computational design and molecular modeling of the interaction of nicotinic acid hydrazide nickel-based complexes with H2S gas†
Abstract
The application of nickel complexes of nicotinic acid hydrazide ligand as a potential gas-sensor and adsorbent material for H2S gas was examined using appropriate density functional theory (DFT) calculations with the ωB97XD/Gen/6-311++G(d,p)/LanL2DZ method. The FT-IR spectrum of the synthesized ligand exhibited a medium band at 3178 cm−1 attributed to ν(NH) stretching vibrations and strong bands at 1657 and 1600 cm−1 corresponding to the presence of ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) and ν(C
O) and ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) vibration modes. In the spectrum of the nickel(II) complex, the ν(C
N) vibration modes. In the spectrum of the nickel(II) complex, the ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) and ν(C
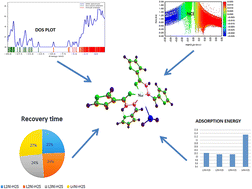
O) and ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) vibration bands experience negative shifts to 1605 cm−1 and 1580 cm−1, respectively, compared to the ligand. This indicates the coordination of the carbonyl oxygen and the azomethine nitrogen atoms to the Ni2+ ion. Thus, the sensing mechanism of the complexes indicated a short recovery time and that the work function value increases for all complexes, necessitating an excellent H2S gas sensor material. Thus, a profound assertion was given that the complex sensor surfaces exhibited very dense stability with regards to their relevant binding energies corresponding to various existing studies.
N) vibration bands experience negative shifts to 1605 cm−1 and 1580 cm−1, respectively, compared to the ligand. This indicates the coordination of the carbonyl oxygen and the azomethine nitrogen atoms to the Ni2+ ion. Thus, the sensing mechanism of the complexes indicated a short recovery time and that the work function value increases for all complexes, necessitating an excellent H2S gas sensor material. Thus, a profound assertion was given that the complex sensor surfaces exhibited very dense stability with regards to their relevant binding energies corresponding to various existing studies.


 Please wait while we load your content...
Please wait while we load your content...