Development of a novel chemoenzymatic route to enantiomerically enriched β-adrenolytic agents. A case study toward propranolol, alprenolol, pindolol, carazolol, moprolol, and metoprolol†
Abstract
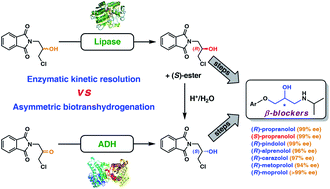
Efficient chemoenzymatic routes toward the synthesis of both enantiomers of adrenergic β-blockers were accomplished by identifying a central chiral building block, which was first prepared using lipase-catalyzed kinetic resolution (KR, Amano PS-IM) as the asymmetric step at a five gram-scale (209 mM conc.). The enantiopure (R)-chlorohydrin (>99% ee) subsequently obtained was used for the synthesis of a series of model (R)-(+)-β-blockers (i.e., propranolol, alprenolol, pindolol, carazolol, moprolol, and metoprolol), which were produced with enantiomeric excess in the range of 96–99.9%. The pharmaceutically relevant (S)-counterpart, taking propranolol as a model, was synthesized in excellent enantiomeric purity (99% ee) via acetolysis of the respective enantiomerically pure (R)-mesylate by using cesium acetate and a catalytic amount of 18-Crown-6, followed by acidic hydrolysis of the formed (S)-acetate. Alternatively, asymmetric reduction of a prochiral ketone, namely 2-(3-chloro-2-oxopropyl)-1H-isoindole-1,3(2H)-dione, was performed using lyophilized E. coli cells harboring overexpressed recombinant alcohol dehydrogenase from Lactobacillus kefir (E. coli/Lk-ADH-Lica) giving the corresponding chlorohydrin with >99% ee. Setting the stereocenter early in the synthesis and performing a 4-step reaction sequence in a ‘one-pot two-step’ procedure allowed the design of a ‘step-economic’ route with a potential dramatic improvement in process efficiency. The synthetic method can serve for the preparation of a broad scope of enantiomerically enriched β-blockers, the chemical structures of which rely on the common α-hydroxy-N-isopropylamine moiety, and in this sense, might be industrially attractive.


 Please wait while we load your content...
Please wait while we load your content...