Effect of the enantiomeric structure of hydrophobic polymers on the encapsulation properties of a second near infrared (NIR-II) fluorescent dye for in vivo deep imaging†
Abstract
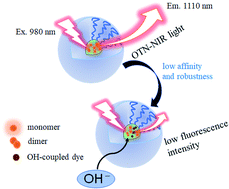
Over-thousand-nanometer (OTN) near-infrared (NIR) fluorophores are useful for biological deep imaging because of the reduced absorption and scattering of OTN-NIR light in biological tissues. IR-1061, an OTN-NIR fluorescent dye, has a hydrophobic and cationic backbone in its molecular structure, and a non-polar counter ion, BF4−. Because of its hydrophobicity, IR-1061 needs to be encapsulated in a hydrophobic microenvironment, such as a hydrophobic core of polymer micelles, shielded with a hydrophilic shell for bioimaging applications. Previous studies have shown that the affinity of dyes with hydrophobic core polymers is dependent on the polarity of the core polymer, and that this characteristic is important for designing dye-encapsulated micelles to be used in bioimaging. In this study, the dye–polymer affinity was investigated using hydrophobic polymer films with different chiral structures of poly(lactic acid). IR-1061 showed higher affinity for L- and D-lactic acid copolymers (i.e., poly(DL-lactic acid) (PDLLA)) than to poly(L-lactic acid) (PLLA), as IR-1061 shows less dimerization in PDLLA than in PLLA. In contrast, the stability of IR-1061 in PDLLA was less than that in PLLA due to the influence of hydroxyl groups. Choosing hydrophobic core polymers for their robustness and dye affinity is an effective strategy to prepare OTN-NIR fluorescent probes for in vivo deep imaging.


 Please wait while we load your content...
Please wait while we load your content...