Electrochemical detection of Cd(ii) ions in complex matrices with nanopipets†
Abstract
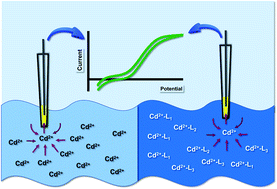
Heavy metal contamination and its detrimental health effects are a growing concern globally. Several metal mitigation systems and regulatory approaches have been implemented to minimize the negative impacts on human health. However, none of these function at maximum efficiency, mainly due to the lack of accurate information about metal speciation. Therefore, there is a critical need to develop novel, cheap, efficient, and robust metal detecting sensors. In this study, we describe the application of a nanopipet based electrochemical sensor to detect aqueous Cd(II) ions. The inner radius of our nanopipets is ∼300 nm, and the fundamental mechanism behind our sensor's response is ion transfer between two immiscible electrolyte solutions (ITIES). The absence of redox behavior makes ITIES an excellent, attractive electrochemical tool to study various ions in aqueous solutions. In this study, we used 1,10-phenanthroline as our ionophore in the organic phase (dichloroethane) to facilitate the transfer of Cd(II) ions from the polar aqueous phase to the less polar organic phase. Unlike previous studies, we characterized our nanopipet in complicated matrices, including, but not limited to, tris buffer and artificial seawater. We performed quantitative assessments to determine our sensor's limit of detection, stability, sensitivity, and selectivity. We further show that our nanosensor can detect free Cd(II) ions in the presence of strong complexing agents such as ethylenediaminetetraacetic acid, 2,3-dimercaptosuccinic acid, etc. We quantified the concentration of free Cd(II) ions in a water sample collected from a local lagoon. Thus, we showcased the power of our nanopipets to act as a robust, accurate, and efficient speciation sensor to detect Cd(II) ions in environmental samples.


 Please wait while we load your content...
Please wait while we load your content...