Asymmetric living supramolecular polymerization of an achiral aza-BODIPY dye by solvent-mediated chirality induction and memory†
Abstract
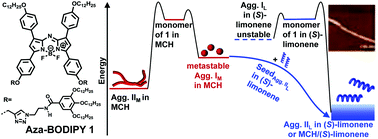
The kinetic self-assembly properties of an achiral aza-BODIPY dye 1 bearing two hydrophobic fan-shaped tridodecyloxybenzamide pendants through 1,2,3-triazole linkages were investigated in detail in chiral solvents (S)- and (R)-limonene by UV/Vis absorption spectroscopy, CD spectroscopy, AFM, and TEM. The spectroscopic studies indicated the formation of thermodynamic H-aggregates (Agg. IIL) with a helical morphology, while metastable aggregates (Agg. IL) could only be observed in the fast cooling process of 1 in limonene. According to further analysis of the spectroscopic data using the nucleation–elongation model, the mechanistic aspects of the self-assembly process of dye 1 were elucidated. The macroscopic chirality and helicity of supramolecular aggregates were found to be dependent on the chirality of the limonene solvents. Since the kinetic trapping of Agg. IL could not be attained in experiments, an effective strategy for asymmetrical living supramolecular polymerization (LSP) of dye 1 was developed by employing the seeds in the chiral solvent limonene and the kinetically-trapped metastable aggregates (Agg. IM) in methylcyclohaxane (MCH). The chiral aggregates Agg. IIL were obtained by multiple-cyclic LSP in the MCH/limonene solvent mixture even at a very low limonene ratio (less than 1%), which could be ascribed to the chiral memory effect of dye 1 aggregates upon continuous dilution of the chiral environment in the solvent mixture during the LSP process.
- This article is part of the themed collection: FOCUS: Macrocyclic and supramolecular chemistry


 Please wait while we load your content...
Please wait while we load your content...