Preferred coordination of polymers at MOFs enables improved lithium-ion battery anode performance†
Abstract
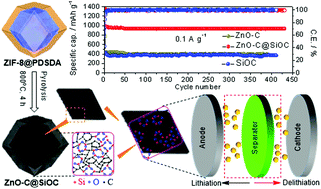
Lithium-ion batteries (LIBs) are widely used energy storage devices due to their low maintenance cost, constant voltage, and high energy density. However, the low coulombic efficiency caused by the side reactions hinders the revival of LIBs. In this work, a facile suspension polymerization method is used to coordinate a linear chain poly[(dimethylsilylene)diacetylene] at ZIF-8, followed by pyrolysis to achieve a controlled architecture of hybrid silicon oxycarbide coated zinc oxide carbon (ZnO–C@SiOC) nanocomposite and improve the poor performance of ZnO–C anode. Notably, the SiOC network generated within an amorphous and stabilized sp2-network not only improves conductivity and active sites but also coordinates with a confined ZnO–C. The confined ZnO–C porous nanoclusters are directly intercalating with the polymer's diacetylene units and alkynyl groups, resulting in an enlarged surface for fast lithium-ion accommodation (8.935 × 10−11 cm−2 s−1) and short ionic diffusion. The ZnO–C@SiOC anode exhibits high reversible capacities of ∼940 mA h g−1 after 430 cycles at 0.1 A g−1 (corresponding to an areal capacity of 2.5 mA h cm−2) and ∼472 mA h g−1 at 0.8 A g−1 with a mass loading of 2.8 mg cm−2. The ultra-fast ability of the ZnO–C@SiOC nanocomposite in a half-cell allows short ionic diffusion and rapid electron transportation, with an extrinsic charge storage pseudocapacitive contribution of 64.16% and a diffusion contribution of 35.86% at 0.4 mV s−1. Furthermore, the ZnO–C@SiOC anode is coupled with a LiFePO4 cathode to assemble a full cell capable of retaining ∼388 mA h g−1 capacity over 180 cycles.


 Please wait while we load your content...
Please wait while we load your content...