Poly(1-halogen-2-phenylacetylenes) containing tetraphenylethene units: polymer synthesis, unique emission behaviours and application in explosive detection†
Abstract
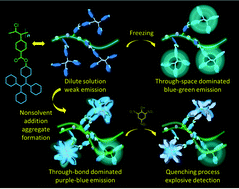
Pd-based catalysts have shown high activity in the polymerization of 1-chloro-2-phenylacetylene monomers under mild conditions, but it is still a challenge to find synthetic routes to achieve polymers with high molecular weight, sufficient solubility and applicable functions. In this work, we report the application of Pd-based catalysts to the preparation of functional poly(1-halogen-2-phenylacetylenes) containing tetraphenylethene (TPE) units. The derived polymer P2 has high molecular weight (Mw over 110 000) and excellent solubility in common organic solvents. Moreover, polymerizable monomers have been extended from 1-chloro-2-phenylacetylene to 1-bromo-2-phenylacetylene derivatives. The introduction of TPE units into the polymer structure furnished the products with aggregation-induced emission (AIE) characteristics. The polymers show weak fluorescence emission in dilute solutions but become highly emissive in concentrated solution and aggregation states. The rigid backbone of the poly(disubstituted acetylene) and the cis-conformer dominant regio-regularity offered by the Pd-based catalyst allow P2 to show dual-band emission features in concentrated solutions and tetrahydrofuran (THF)/water mixtures with high water fractions (fw). These two bands, which are referred to as λ400 and λ470 bands, are ascribed to the through-bond conjugation of TPE units and the intrachain excimers of the polymer backbone, respectively. The intensity of the λ400 band increases very fast when the concentration of P2 in THF increased over 10−4 M or fw increased from 20% to 80% in THF/water mixtures, but the intensity of the λ470 band only has small enhancement. These emission behaviors allow us to employ P2 as a ratiometric fluorescent probe in chemical detection. With picric acid (PA) as a model explosive, a super-quenching process has been observed. The limit of detection is 63 nM and the quenching constant (KSV) calculated from the Stern–Volmer equation is high up to 55 000 M−1 when the concentration of PA is in the range of 10–50 μM.


 Please wait while we load your content...
Please wait while we load your content...