Ionic liquid and lysine co-assisted synthesis of the highly dispersed Ni/SAPO-11 catalyst†
Abstract
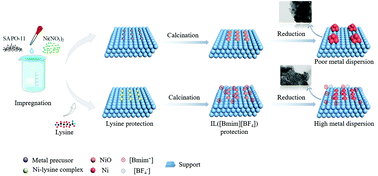
The synthesis of highly-dispersed non-noble metal based catalysts and their application in hydrogenation/dehydrogenation still remain challenging. Therefore, it is of great importance to protect metals from aggregation in both calcination and reduction processes. In this research, density functional theory (DFT) calculations were used to elucidate the stabilization effect of ionic liquid [Bmim][BF4] on Ni in the reduction process, showing that [Bmim][BF4] could efficiently inhibit Ni aggregation on the support. By further analysis combined with theoretical calculations, we found that the degree of metal dispersion before reduction directly determines the effect of [Bmim][BF4]. Guided by the DFT results, the method of co-assisted preparation of Ni/SAPO-11 using [Bmim][BF4] and lysine was adopted. Lysine forms a coordination compound with nickel oxides. The steric hindrance generated by the Ni–lysine complex can effectively inhibit the aggregation of nickel oxides during the calcination process. The DFT calculations revealed that during the reduction process, [BF4−] plays a dominant role in stabilization and electronic modulation by directly interacting with Ni on the support. The combination effects of [Bmim][BF4] and lysine effectively inhibit metal aggregation, leading to ultra-small nickel particles with a mean size of 4.4 nm over the obtained catalyst. Furthermore, the presence of [Bmim][BF4] also tunes the electronic state of Ni species. Using hexane isomerization as a probe reaction, the result indicates the excellent catalytic activity of the obtained catalyst (≈93.7% isomer selectivity at 63.5% conversion), much superior to that of Ni/SAPO-11 obtained from the traditional impregnation route (≈75.8% isomer selectivity at 64.6% conversion). This work provides a new method for the preparation of highly effective non-noble metal supported catalysts.
- This article is part of the themed collection: Synthesis, modification and tailoring of properties of nanoporous materials


 Please wait while we load your content...
Please wait while we load your content...