Structural dimension modulation in a new oxysulfide system of Ae2Sb2O2S3 (Ae = Ca and Ba)†
Abstract
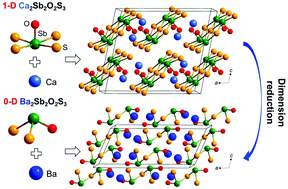
Inspired by the abundant structural diversity and potential applications of Sb-based oxysulfides, two new compounds with the same stoichiometry, Ae2Sb2O2S3 (Ae = Ca, Ba), were successfully synthesized via solid state reactions. Both crystallize in the C2/c space group (no. 15) of a monoclinic system, however, possess different structural dimensions induced by different sizes of template alkaline-earth ions. Ca2Sb2O2S3 is composed of one-dimensional anionic [Sb2O2S3]4− chains isolated by Ca2+ ions, while Ba2Sb2O2S3 consists of zero-dimensional anionic [Sb2O2S3]4− clusters separated by Ba2+ ions. Higher structural dimension endows the 1D Ca2Sb2O2S3 with a higher melting point (m.p. = 720 °C) and a narrower band gap (Eg = 2.36 eV), compared with the m.p. = 692 °C and Eg = 2.78 eV of Ba2Sb2O2S3. Both exhibit interesting photoluminescence properties with multiemission characteristics. DFT calculations reveal that the band edges of Ae2Sb2O2S3 are mainly composed of Sb−S bond orbitals, while O 2p orbitals also contribute to the valence band maximum of Ba2Sb2O2S3.
- This article is part of the themed collection: FOCUS: Metal and Metal-Containing Clusters


 Please wait while we load your content...
Please wait while we load your content...