Facile and efficient photocatalyst for degradation of chlortetracycline promoted by H2O2†
Abstract
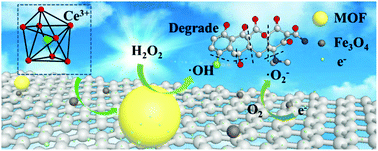
The composite photocatalyst based on a cerium(III) metal–organic framework (MOF-1 or 1), graphene oxide (GO), and Fe3O4 was constructed for the first time and was investigated for the degradation of chlortetracycline. A superior synergistic effect of graphene oxide and MOF-1 was achieved for improving photocatalysis. The optimal composite (1/GO/Fe3O4) with 9.0 wt% graphene oxide displayed the highest photocatalytic performance in the degradation of chlortetracycline with the help of hydrogen peroxide (H2O2), with a removal rate of 80.5% in 180 minutes, which exhibited much higher photocatalytic performance than that of parent MOF-1. The enhanced photocatalytic performance was mainly attributed to the modification of graphene oxide, which can inhibit the recombination of the photogenerated carrier as an electron transporter and improve the absorption of visible light. Furthermore, the composite displayed superparamagnetic behavior that permitted the magnetic separation and convenient recovery of the photocatalyst from the reaction mixture. Importantly, the photocatalyst exhibited high stability and could be used recurrently. The photocatalytic mechanism involved the excitation of electrons from the VB to the CB in MOF-1 excited by visible light irradiation, the transport of electrons between MOF-1 and graphene oxide, and the generation of active groups (˙OH and ˙O2−).


 Please wait while we load your content...
Please wait while we load your content...