Cu/Co/CoS2 embedded in S,N-doped carbon as highly efficient oxygen reduction and evolution electrocatalyst for rechargeable zinc–air batteries†
Abstract
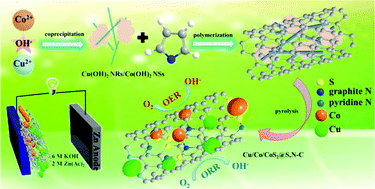
In order to improve the retarded oxygen reduction and evolution reaction (ORR/OER) in rechargeable metal–air cells in electrochemical energy conversion systems, constructing multiphase nanostructured catalysts is an alternative strategy, where the interface between different phases can be enriched and synergistic interaction can be realized. Here, a scalable one-pot approach is designed to achieve Cu/Co/CoS2@S,N-doped carbon using two-phase metal hydroxides (Cu(OH)2 nanorods/Co(OH)2 nanosheets) with polypyrrole on their surface as precursors followed by high-temperature carbonization. The introduction of multiphase interfaces affects their charge states and facilitates the charge transport processes. Meanwhile, the embedded structure protects the Cu/Co/CoS2 from self-aggregation and corrosion. As a result, the product performs satisfactory oxygen reduction activity and stability with a half-wave potential of 0.811 V vs. RHE and a current density of 7.03 mA cm−2. Meanwhile, it exhibits an overpotential of 445 mV at 10 mA cm−2 for oxygen evolution. The rechargeable Zn–air battery composed of Cu/Co/CoS2@S,N–C as the cathode material achieves a maximum power density of 110.5 mW cm−2 and a low charge/discharge voltage gap of 0.87 V, even after 80 h at 5 mA cm−2.


 Please wait while we load your content...
Please wait while we load your content...