Facile construction of functional poly(monothiocarbonate) copolymers under mild operating conditions†
Abstract
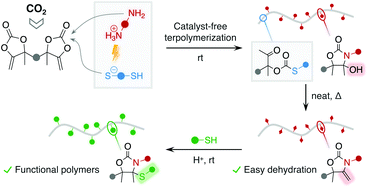
The installation of both oxazolidone and thiocarbonate linkages within a single polymer backbone remains elusive by simple procedures under mild conditions. In this work, we report the construction of copolymers containing these two linkages by the one-pot terpolymerization of diamines, dithiols and a CO2-sourced activated dicyclic carbonate of the α-alkylidene type. This process is facile and efficient with many commercially available diamine and dithiol comonomers, and occurs at room temperature under catalyst-free conditions. In contrast to polyoxazolidones that are insoluble in many organic solvents, the copolymers are highly soluble, facilitating their processing as adhesives. The polymers present an unusual thermal behavior with the presence of two glass transition temperatures. Importantly, they can undergo facile and quantitative dehydration by a simple thermal treatment at moderate temperature (110–160 °C) without using any catalyst or dehydrating agent, thereby furnishing novel polymers presenting both thiocarbonate and α-alkylidene oxazolidone linkages. The pendant double bonds are exploited for the facile thiol–ene functionalization with a scope of commercially available thiols under mild conditions. This work gives access to functional poly(monothiocarbonate)-based copolymers under mild operating conditions without requiring specific equipment.
- This article is part of the themed collection: Chalcogen-containing polymers


 Please wait while we load your content...
Please wait while we load your content...