Efficient suppression of the chain transfer reaction in ethylene coordination polymerization with dibenzosuberyl substituents†
Abstract
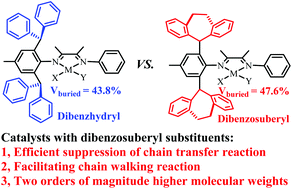
The chain transfer reaction plays an important role in governing the polymer molecular weight in coordination polymerization. In this work, we demonstrate that dibenzosuberyl substituents are efficient in suppressing chain transfer reactions and significantly enhancing the molecular weight of polymers and copolymers in ethylene (co)polymerization with α-diimine Ni(II) and Pd(II) species. Herein, a family of α-diimine Ni(II) and Pd(II) complexes with dibenzosuberyl substituents were designed, synthesized and characterized. In the nickel-catalyzed ethylene polymerization, dibenzosuberyl Ni(II) catalysts exhibited high activities (level of 106 g mol−1 h−1) and yielded highly branched (80–93/1000C) polyethylenes with high molecular weights (up to 586.4 kg mol−1). On the other hand, in the corresponding palladium-catalyzed ethylene polymerization, the Pd(II) catalysts displayed moderate activities (level of 105 g mol−1 h−1) and yielded highly branched (72–113/1000C) polyethylenes with high molecular weights (up to 297.3 kg mol−1). Moreover, dibenzosuberyl Pd(II) catalysts generated highly branched (72–107/1000C) E-MA copolymers with high molecular weights (up to 78.5 kg mol−1) and high incorporation ratios (up to 9.6 mol%). As compared with the classical dibenzhydryl Ni(II) and Pd(II) catalysts, dibenzosuberyl catalysts displayed an outstanding ability to suppress chain transfer reactions and generated polymers and copolymers with 1–2 orders of magnitude higher molecular weights during ethylene (co)polymerization. The sandwich-like structure formed by dibenzosuberyl substituents was proposed to be responsible for the observed phenomenon.


 Please wait while we load your content...
Please wait while we load your content...