Influence of the molecular weight and size distribution of PSS on mixed ionic-electronic transport in PEDOT:PSS†
Abstract
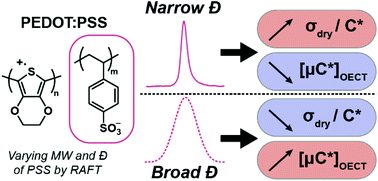
The commercially available polyelectrolyte complex poly(3,4-ethylenedioxythiophene):poly(styrene sulfonate) (PEDOT:PSS) is ubiquitous in organic and hybrid electronics. As such, it has often been used as a benchmark material for fundamental studies and the development of new electronic devices. Yet, most studies on PEDOT:PSS have focused on its electronic conductivity in dry environments, with less consideration given to its ion transport, coupled ionic-electronic transport, and charge storage properties in aqueous environments. These properties are essential for applications in bioelectronics (sensors, actuators), charge storage devices, and electrochromic displays. Importantly, past studies on mixed ionic-electronic transport in PEDOT:PSS neglected to consider how the molecular structure of PSS affects mixed ionic-electronic transport. Herein, we therefore investigated the effect of the molecular weight and size distribution of PSS on the electronic properties and morphology of PEDOT:PSS both in dry and aqueous environments, and overall performance in organic electrochemical transistors (OECTs). Using reversible addition–fragmentation chain transfer (RAFT) polymerization with two different chain transfer agents, six PSS samples with monomodal, narrow (Đ = 1.1) and broad (Đ = 1.7) size distributions and varying molecular weights were synthesized and used as matrices for PEDOT. We found that using higher molecular weight of PSS (Mn = 145 kg mol−1) and broad dispersity led to OECTs with the highest transconductance (up to 16 mS) and [μC*] values (∼140 F cm−1 V−1 s−1) in PEDOT:PSS, despite having a lower volumetric capacitance (C* = 35 ± 4 F cm−3). The differences were best explained by studying the microstructure of the films by atomic force microscopy (AFM). We found that heterogeneities in the PEDOT:PSS films (interconnected and large PEDOT- and PSS-rich domains) obtained from high molecular weight and high dispersity PSS led to higher charge mobility (μOECT ∼ 4 cm2 V−1 s−1) and hence transconductance. These studies highlight the importance of considering molecular weight and size distribution in organic mixed ionic-electronic conductor, and could pave the way to designing high performance organic electronics for biological interfaces.
- This article is part of the themed collections: 2025 Journal of Materials Chemistry Lectureship shortlisted candidates, Polymer Chemistry 15th anniversary regional spotlight collection: The Americas, Polymer Chemistry Most Popular 2022 and Tailoring dispersity and shape of molecular weight contributions


 Please wait while we load your content...
Please wait while we load your content...