Redox activated amines in the organophotoinduced alkylation of coumarins†
Abstract
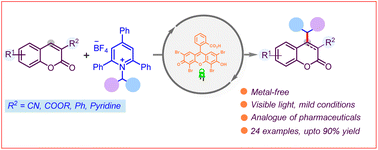
The coumarin core represents the quintessential scaffold of many natural products. While C-3 alkylation is easily achievable, effective greener strategies for C-4 alkylation have been less forthcoming. Herein, we report a metal-free photoinduced deaminative strategy for C-4 alkylation of coumarins using redox activated secondary and benzylic amine derived Katritzky pyridinium salts.


 Please wait while we load your content...
Please wait while we load your content...