Total synthesis of (±)-mersicarpine following a 6-exo-trig radical cyclization†
Abstract
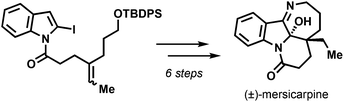
Described is a total synthesis of racemic mersicarpine from diethyl 4-oxopimelate. The synthetic route takes advantage of a 2-indolyl radical cyclization to construct the pyrido[1,2-a]indole scaffold bearing the all-carbon quaternary stereocenter.
- This article is part of the themed collection: New Talent


 Please wait while we load your content...
Please wait while we load your content...