A concise synthesis of herbertenolide†
Abstract
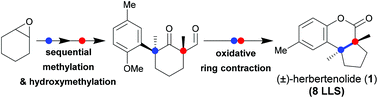
A concise synthesis of (±)-herbertenolide has been accomplished herein. The strategy relies on a H2O2-mediated oxidative ring contraction of all-substituted cyclic α-formyl ketones for the stereospecific construction of contiguous quaternary carbon centers (CQCCs). Furthermore, a Sc(OTf)3/chiral N,N′-dioxide catalyzed asymmetric Michael addition of benzofuranone to MVK has been optimized for forging a chiral aromatic quaternary carbon center, which enables the formal synthesis of (+)-ent-herbertenolide.


 Please wait while we load your content...
Please wait while we load your content...