Capture and electrochemical conversion of CO2 in molten alkali metal borate–carbonate blends†
Abstract
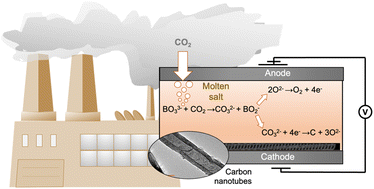
A family of blended compositions of molten mixed lithium and sodium borate (Li1.5Na1.5BO3) and eutectic lithium–potassium carbonate (Li1.24K0.76CO3) salts has been introduced as reversible carbon dioxide absorbents and as media for CO2 electrolysis for carbon conversion. Material properties, temperature effects and kinetics of CO2 uptake were examined. Li, Na borate can absorb up to 7.3 mmol g−1 CO2 at 600 °C. The blended borate–carbonate compositions are molten in the 550–600 °C temperature range, with viscosity adjustable to within a 10–1000 Pa s window depending on the borate/carbonate ratio. The blends can withstand cyclic temperature and CO2 pressure swings without significant deterioration of their CO2 uptake capabilities. Addition of eutectic carbonate into mixed Li, Na borate salts lowers overall CO2 uptake due to the lower solubility of CO2 in carbonate. However, addition of the eutectic lowers the temperature of the pressure swing operation and dramatically accelerates the CO2 uptake during the initial stage of the absorption, potentially enabling a faster cycling. Electroreduction of CO2 and carbon deposition on a galvanized steel cathode was more effective with increasing carbonate fraction in the molten alkali borate/carbonate blend. Blended borate/carbonate compositions with 50–60% borate content possessed sufficiently high loading capacity for CO2 and simultaneously enabled maximum carbon product yield and Coulombic efficiency. Most of the recovered carbon product was shown to be in the form of multiwalled carbon nanotube.
- This article is part of the themed collection: CO2 capture and conversion


 Please wait while we load your content...
Please wait while we load your content...