SO3 decomposition over silica-modified β-SiC supported CuFe2O4 catalyst: characterization, performance, and atomistic insights†
Abstract
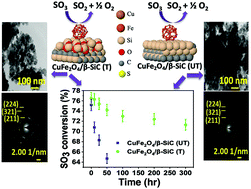
The sulfur–iodine (S–I) thermochemical water-splitting cycle is one of the potential ways to produce hydrogen on a large scale. CuFe2O4 was dispersed over modified silica or treated β-SiC and untreated β-SiC using the wet impregnation method for SO3 decomposition, which is the most endothermic reaction of the S–I cycle. Various state-of-the-art techniques such as XRD, FT-IR, BET, XPS, TEM, HR-TEM, FESEM-EDS and elemental mapping were employed to characterize both the synthesized catalysts. CuFe2O4 catalyst supported on silica-modified β-SiC resulted in enhanced catalytic activity and stability due to better metal-support interaction. In order to get a better insight into the reaction mechanism over this bimetallic catalyst, the first principles based simulation under the framework of density functional theory was performed. We have found that the presence of Cu gives rise to an improved charge localization at the O-vacancy site alongside favourable reaction kinetics, which results in an enhanced catalytic activity for the CuFe2O4 nano-cluster compared to that of a single metallic catalyst containing Fe2O3 nano-cluster.


 Please wait while we load your content...
Please wait while we load your content...