Fe and P dual-doped nickel carbonate hydroxide/carbon nanotube hybrid electrocatalysts for an efficient oxygen evolution reaction†
Abstract
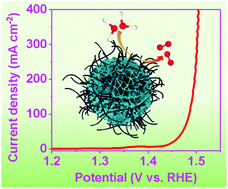
Designing cost-effective and highly active oxygen evolution reaction (OER) electrocatalysts is critical for large-scale hydrogen production from electrocatalytic water splitting. Herein, Fe and P dual-doped nickel carbonate hydroxide/carbon nanotubes (Fe, P–NiCH/CNTs) were fabricated through a solvothermal method. By virtue of the optimized electronic structure, improved conductivity and enriched active sites, the as-fabricated Fe, P–NiCH/CNT hybrid electrocatalyst exhibits superior OER activity, with a low overpotential of 222 mV at 20 mA cm−2 and robust durability, confirming its potential as a highly efficient OER electrocatalyst. Moreover, theoretical calculations demonstrate that the doped Fe and surface adsorbed PO43− can regulate the electronic structure of evolved NiOOH and decrease the energy barrier of the rate-determining step, thus leading to improved OER activity. The strategy presented in this work can also be employed to fabricate other transition metal carbonate hydroxides for various electrocatalytic applications.


 Please wait while we load your content...
Please wait while we load your content...