Silybin and its congeners: from traditional medicine to molecular effects†
Abstract
Covering: 2015 up to 2022 (Feb)
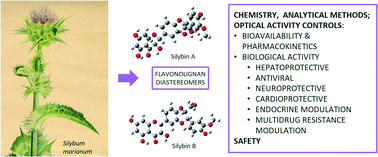
Silymarin, an extract of milk thistle (Silybum marianum) fruits, has been used in various medicinal applications since ancient times. A major component of silymarin is the flavonolignan silybin and its relatives isosilybin, silychristin, silydianin, 2,3-dehydrosilybin, and some others. Except for silydianin, they occur in nature as two stereomers. This review focuses on recent developments in chemistry, biosynthesis, modern advanced analytical methods, and transformations of flavonolignans specifically reflecting their chirality. Recently described chemotypes of S. marianum, but also the newest findings regarding the pharmacokinetics, hepatoprotective, antiviral, neuroprotective, and cardioprotective activity, modulation of endocrine functions, modulation of multidrug resistance, and safety of flavonolignans are discussed. A growing number of studies show that the respective diastereomers of flavonolignans have significantly different activities in anisotropic biological systems. Moreover, it is now clear that flavonolignans do not act as antioxidants in vivo, but as specific ligands of biological targets and therefore their chirality is crucial. Many controversies often arise, mainly due to the non-standard composition of this phytopreparation, the use of various undefined mixtures, the misattribution of silymarin vs. silybin, and also the failure to consider the chemistry of the respective components of silymarin.


 Please wait while we load your content...
Please wait while we load your content...