Atomic and nanosized Co species functionalized N-doped porous carbon hybrids for boosting electrocatalytic oxygen reduction†
Abstract
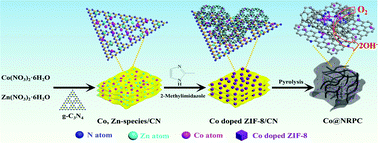
Realizing the synergistic interaction of catalytically active sites with carbonaceous substrates plays an essential role in the development of high-performance carbon-based oxygen reduction reactions (ORRs) for Zn–air batteries (ZABs). In this work, a facile synthesis strategy is engineered to construct high-efficiency ORR electrocatalysts in which atomic CoNx moieties and nanosized metallic Co are integrated with N-doped porous carbon (denoted as Co@NRPC) by pyrolyzing hybrid precursors consisting of Co-doped zeolitic imidazolium frameworks (ZIFs) and graphitic carbon nitride (g-C3N4). The N-doped porous carbon substrate is composed of interconnected nanopolyhedra, nanotubes and nanosheets. The synergistic interaction of multiple active components with a hierarchically porous N-doped carbon substrate endows the Co@NRPC electrocatalysts with more accessible active sites, favorable electrical conductivity, enhanced mass transport and stability. As a result, the optimized Co@NRPC-90 features an impressive ORR performance with a high half-wave potential of 868 mV (vs. the reversible hydrogen electrode), satisfactory stability and methanol tolerance, which are superior to those of the commercial Pt/C electrocatalyst. Furthermore, the as-assembled solid-state ZAB with Co@NRPC-90 as an air-cathode electrocatalyst delivers a competitive power density (184 mW cm−2) and specific capacity (1610 mA h g−1Zn) much higher than those of a ZAB based on a commercial Pt/C electrocatalyst (136 mW cm−2 and 1034 mA h g−1Zn).


 Please wait while we load your content...
Please wait while we load your content...