Tuning of energy levels, transport properties and device performance of naphthalenediimide derivatives by introduction of Michael addition reaction†
Abstract
The current work describes the Michael addition (MA) reaction between amine group appended NDI derivatives and α,β-unsaturated ketones (chalcones) with an aim to tune the optical and electrochemical properties of NDI derivatives via an easy and scalable synthesis method. Two NDI derivatives having aromatic (1) and aliphatic (2) amine N-imide substituents were synthesized and further used for the synthesis of MA products. The products were evaluated for their optical, electrochemical and transport properties. Aggregation behavior of the molecules was studied and established by absorption and emission spectroscopy. It is observed that NDIs and their respective MA derivatives (3 and 4) show strong aggregation behavior. Most interestingly the HOMO–LUMO energy levels in MA products were uplifted which is generally not achieved by imide-N substitutions. The driving force for intramolecular electron transfer 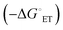 was calculated from their first oxidation and reduction potentials indicating the formation of charge separated states. MA products also showed improved electron mobility compared to their NDI precursors. Intramolecular charge transfer interaction was established by fluorescence and femtosecond transient absorption spectroscopy (TAS). The materials have been studied for their use as electron transport layer (ETL) materials in inverted organic solar cells and an efficiency of 3% has been obtained. Overall, Michael addition reaction on NDI has resulted in effective tuning of optical properties, energy levels and transport properties without involving tedious multistep core substitution reactions.
was calculated from their first oxidation and reduction potentials indicating the formation of charge separated states. MA products also showed improved electron mobility compared to their NDI precursors. Intramolecular charge transfer interaction was established by fluorescence and femtosecond transient absorption spectroscopy (TAS). The materials have been studied for their use as electron transport layer (ETL) materials in inverted organic solar cells and an efficiency of 3% has been obtained. Overall, Michael addition reaction on NDI has resulted in effective tuning of optical properties, energy levels and transport properties without involving tedious multistep core substitution reactions.



 Please wait while we load your content...
Please wait while we load your content...