The synergistic effect of PMS activation by LaCoO3/g-C3N4 for degradation of tetracycline hydrochloride: performance, mechanism and phytotoxicity evaluation†
Abstract
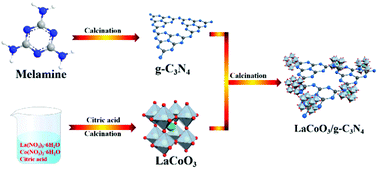
Tetracycline hydrochloride (TC) is a typical refractory organic pollutant, and is frequently detected in different water environments, which will increase microbial drug resistance, destroy ecosystems and threaten human health. It is urgent to develop a simple and effective catalyst for TC removal. In this paper, a facile two-step calcination method was used to prepare a LaCoO3/g-C3N4 composite catalyst with high activity and high stability for PMS activation to degrade TC. Various characterization results confirmed that the LaCoO3/g-C3N4 composite catalyst possessed smaller particle size, higher stability and faster electronic transmission ability. Benefiting from the synergistic effect between LaCoO3 and g-C3N4, the LaCoO3/g-C3N4 catalyst showed high activity for PMS activation to degrade different antibiotics. Furthermore, the results of the quenching experiments and the electron paramagnetic resonance (EPR) tests showed that the generated SO4˙− and 1O2 radicals during the PMS activation process played a key role in pollutant degradation. The effect of different reaction conditions (including pH, temperature, PMS concentration, catalyst dosage and different anions) on the performance of the catalyst was also investigated. The catalyst cycle evaluation experiments showed that the LaCoO3/g-C3N4 composite catalyst has high stability and reusability. The phytotoxicity evaluation based on seed germination experiments indicated that the LaCoO3/g-C3N4 composite catalyst system can effectively reduce the toxicity of antibiotics.


 Please wait while we load your content...
Please wait while we load your content...