Magnetically recyclable core–shell MOF nanoparticles of Fe3O4@PDA@UIO-66-NH2 grafted by organic acids for intensified cationic dye adsorption†
Abstract
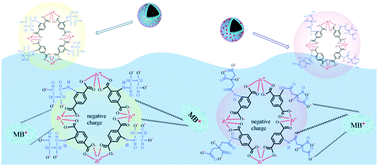
Two kinds of hierarchical magnetic metal–organic framework (MOF) nanoparticles Fe3O4@PDA@UIO-66-NH-tar (mUIO-66-NH-tar) and Fe3O4@PDA@UIO-66-NH-citr (mUIO-66-NH-citr) were fabricated through post-synthetic modification. The novel nanoparticles were then characterized using SEM, FTIR, PXRD, XPS and surface area analysis. Owing to the porous structure and the high density of the carboxylic acid group (primary sorption sites), the as-prepared adsorbents demonstrated a very fast adsorption rate and a much higher adsorption capacity of methylene blue (MB, as a model of cationic dyes) from aqueous solutions in an expanded pH range. The highest adsorption capacities of mUIO-66-NH-tar and mUIO-66-NH-citr were 381.7 and 432.9 mg g−1, respectively. Experimental results indicated that the pseudo-second order rate equation and Langmuir isotherm model described the kinetics and adsorption isotherm of MB accurately. At higher solution pH (pH > pHpzc of adsorbents), the adsorption of MB by the two adsorbents was mainly driven by electrostatic attraction (EA), hydrogen bonding (HB) and π–π stacking, whereas cation exchange might be the predominant adsorption mechanism at lower solution pH (pH < pHpzc of adsorbents). Moreover, both as-prepared nanoparticles were observed to have high reusability, which could be used over six adsorption–desorption cycles without significant capacity loss.


 Please wait while we load your content...
Please wait while we load your content...